 
The pore size and the network structure of the gel provide an additional sieving mechanism by retarding the motion of particles/molecules that increases the resolving power of this separation technique. This helps to eliminate thermal convection forces in the liquid medium, which would interfere with the separation process. Gels that have porous network structures allow the movement of solvent and solute particles through the pores. In addition to the size and charge, the rate of particle movement depends on its shape and the density of the gel through which it moves. It should be noted that the above equations deal with spherical particles. In gel electrophoresis, the particles to be separated are placed in wells of a gel medium, and an electric field is applied to the medium, which results in separated bands of particles. This forms the basis of the separation of charged particles via electrophoresis. Since under an electric field, the directions and rates of motion of particles in a medium vary depending on their charges and charge densities, the application of the electric field for a given period of time could result in completely different displacements for different particles. 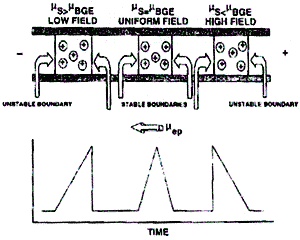 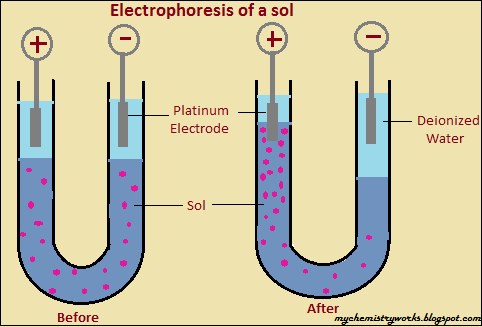
The direction of particle movement under an applied field depends on the nature of the electric charge on the particle (positively charged particles to the cathode and the negatively charged ones to the anode). Thus the electrophoretic mobility in a given medium varies depending on the charge density of the particles. Q/Rs is also known as the charge density of the particle. The above equation shows that the electrophoretic mobility, U, is directly proportional to the charge (Q) and inversely proportional to the particle size (Rs) and the coefficient of viscosity (?) of the medium. For a spherical particle the frictional coefficient is given by f = 6 £S £bRs, where £b is i P is the coefficient of viscosity and Rs (Stokes radius) is the radius of the hydrated sphere.įrom the above equations, one obtains f v = QE, or U (electrophoretic mobility) = v/E = Q/f = Q/6 £S £bR s, The frictional coefficient, f, depends on the size and shape of the molecule. The frictional force is given by, F fr = -f v, where f is a constant called the frictional coefficient and v is the velocity of the particle. Under this condition, if the particle with charge, +Q, moves at a constant velocity (the acceleration, a, of the particle is 0) toward the cathode, the net force Fnet on the particle is zero (since F = ma). 
Two opposing forces act on the particle: the force exerted by the field, FE = QE, in the direction of the movement toward the cathode, and the frictional force, Ffr, retarding its motion toward the cathode. Suppose a particle with charge, +Q, is moving toward the negative electrode (cathode) in a nonconducting medium under an electric field, E. This has also been used for the size separation (based on mass to charge ratio) of nanoclusters and nanoparticles. Gel electrophoresis technique is often used in biotechnology and forensic laboratories for the separation of biological macromolecules such as DNA, RNA, protein, etc. These days electrophoresis is often conducted by using agar or polyacrylamide gels as media, and the technique is known as 'gel electrophoresis. The electrophoresis phenomenon provides an analytical technique for the separation of charged particles/molecules by applying an electric field. Electrophoresis of positively charged particles or cations is termed cataphoresis, and similarly, the electrophoresis of negatively charged particles or anions is termed anaphoresis. Ferdinand Frederic Reuss reported observation of this phenomenon for the first time in 1807. It involves the movement of charged dispersed particles with respect to the dispersion medium under the influence of a spatially uniform electric field. Electrophoresis is an electrokinetic phenomenon. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
